News
Latest News
Future Today Redesigns Streaming App Interfaces
By Jon Lafayette published
Change designed to increase consumer engagement, brand interaction
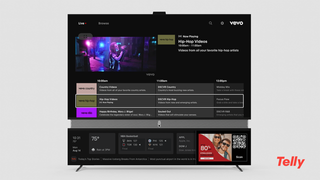
Vevo Brings Music Videos To Telly’s Dual Screen TVs
By Jon Lafayette published
Set, free to consumers, also feature six-speaker sound systems

Comcast Earnings Flat As Video, Broadband Sub Losses Continue
By Jon Lafayette published
Peacock adds 3 million subscribers, cuts losses

TelevisaUnivision 1Q Loss Widens But DTC Business Expected To Turn Profit in Second Half
By Jon Lafayette published
U.S. advertising sales edge up

'Problematic' FCC Conditions On Station Sale Could Create Detroit Drama
By Jon Lafayette published
WADL station owner expects transaction to proceed and give the CW an affiliate in the market, while commissioner Brendan Carr says application was ‘denied’ by Media Bureau

Attend 'The Business of TV News' Event in D.C. on May 2
By Michael Malone published
Martha Raddatz, Shannon Bream, Glenn Kirschner set to deliver keynotes

Sinclair Names Kevin Coskren Chief Meteorologist at KTUL Tulsa
By Jon Lafayette published
Joins ABC affiliate from AccuWeather Network

Paramount Plus Renews ‘Dora’
By Michael Malone published
Spinoff of ‘Dora the Explorer’ debuted earlier this month
Broadcasting & Cable Newsletter
The smarter way to stay on top of broadcasting and cable industry. Sign up below